Introduction to Gas Chromatography for New and Advanced Users – Carrier Gas
Types of Carrier Gas
Carrier gases in gas chromatography play a crucial role by transporting (carrying) the vaporized sample molecules through the column to the detector. The carrier gas enhances the cost-efficiency and speed of gas chromatography compared to liquid chromatography. The carrier gas must be an inert gas, carrying the sample without interacting with the compounds in the sample. Examples include helium (He), nitrogen (N₂), hydrogen (H₂), and argon (Ar). He and N₂ have been the most commonly used gases to date, with H₂ playing a bigger role nowadays.
Recently, we observe a massive increase in the use of H₂ as a carrier gas due to the potential future shortage of helium and extensive runtimes when using nitrogen as a carrier gas. In capillary columns, He is preferred because it maintains the separating resolution at high linear velocity (the speed at which the sample travels through the column), which we will explain later in our blog. Refer to table 1 below for a comparison of the three most commonly used carrier gases.
Table 1. Various carrier gases comparison
| Carrier gas | Pros | Cons |
| Helium
(He) |
Inert and not flammable
Gives high resolution |
Expensive
Not easily available |
| Hydrogen (H2) | High diffusivity
High linear velocity range Good separation efficiency Shorter run times = shorter analysis |
Dangerous – flammable
Not completely inert Can strip dirt from ion source Can react with some compounds at higher temperatures |
| Nitrogen
(N2) |
Cheap and readily available | Not suitable for temperature programmable GC analysis
Lower or poor compounds separation Longer (long) analysis time |
Gas Chromatography Seperation
Carrier gas always flows into the detector therefore it is necessary to use one with a high purity (99.995 % or higher). Carrier gases with high purity can help to suppress baseline noise. Low purity gases will cause massive problems especially when using Mass Spectrometry detectors connected to gas chromatography systems due to increased content of impurities and water. Refer to table below for recommended gas purities when using gas chromatography.

When GC was first developed, the primary mechanism to control and direct the gas flow (carrier gas) within the instrument was by controlling the pressure only. For convenience, the initial pressure (sometimes known as head pressure) was fixed and all the methods created were based on constant pressure mode. There is, however, a major issue in gas chromatography: peak broadening, which needs to be discussed.
There are 3 different carrier gas flow control modes that are commonly used in gas chromatography. We can look directly into the associated units to compare the three control modes:
- constant pressure mode: Pa (controls pressure of carrier gas)
- constant flow mode: cm³/s (volume of gas delivered every second)
- constant linear velocity mode: cm/s (distance which injected sample travels every second)
In constant pressure mode, the head pressure remains fixed. As the sample travels further from the injection port, it experiences less force and slows down as column temperature increases and the gas phase relaxes. This results in peak broadening with increased retention time. This impact is more significant in capillary columns, where the column length is much longer than in packed columns.
Over time, GC methods have shifted from the above carrier gas control mode to improve chromatographic performance.
Both constant flow and linear velocity modes control gas movement similarly. They both overcome the weakness of constant pressure mode by correcting the volume passing through a column or the distance traveled down the column every second. However, columns can expand slightly at higher temperatures during temperature gradient chromatography. The difference between these two modes becomes significant when the column potentially changes its diameter slightly.
In an ideal situation, when a column (tube) has a smaller diameter, the same volume occupies a greater length of the column. Let’s assume 5 cm (blue marker/cylinder in Figure 1 below). For the same volume to pass points 1 and 2 in a given time (Figure 1), the speed must be greater at point 2.
Figure 2. Fluid Flows Through a narrower column faster: The same volume of fluid occupies a different cross-sectional area (A) at points 1 and 2, where the column size is different. This causes a different flow rate, and thus a different linear velocity (v).
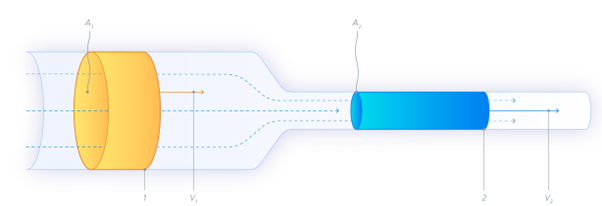
The process is exactly reversible. If the fluid flows in the opposite direction, its speed will decrease when the tube widens. This “speed” is what we know as the linear velocity. During analysis, with the heating program of the GC oven, capillary columns expand gradually. As a result, if the constant flow mode is used, linear velocity will slow down progressively. This affects the separation efficiency.
There is another situation where the constant linear velocity mode comes in handy. When the tail pressure is lower (i.e. vacuum), the flow will be faster than with higher tail pressure (i.e. atmospheric pressure). As a result, the retention time between a GC with an atmospheric detector like FID and a GC with a detector in a vacuum state like mass spectrometer (MS) cannot be matched interchangeably. The issue can be resolved if the constant linear velocity mode is employed. In this mode, carrier gas flows through the column regardless of the detector pressure. What is controlled is the speed through the section of the column. In constant linear velocity mode, it is the same regardless of the column diameter.
Regarding ease of use and complexity of the carrier gas flow modes:
- Pressure mode is easiest to maintain – it is easier to control the pressure, as you only need a regulator to do that
- Flow mode is more sophisticated to control – you need to implement some correction factors, a feedback mechanism which will adjust the pressure regularly so that the volume passing through a column is maintained
- Linear velocity mode is most complex to control – additional corrections are needed for the cross-sectional area are needed and electronics need a more sophisticated calculation algorithm
The biggest advantage of linear velocity control mode is the consistency of this flow mode regardless of the GC instrument brand you use. It is the most universal flow mode control. It maintains the optimal resolution efficiency of chromatographic separation you expect from GC. Across multiple instruments, constant linear velocity ensures the reproducibility of results. It is the best mode for GC methods development. It is very easy to transfer between instruments and laboratories. This is true even for reproducibility between vacuum detectors and ambient pressure detectors.
Many GC detectors work best with only certain types of carrier gases. For instance, when working with packed columns and FID (flame ionization detector), ECD (electron capture detector) or TCD (thermal conductivity detector) the carrier gas usually is nitrogen (N2).
On the other hand, when working with capillary columns and FID or TCD, helium (He) is the carrier gas of choice. A combination of capillary column with ECD detector favors nitrogen or argon-methane mixtures as carrier gases.
Gas Chromatography – Master the Basics
I hope that our second 2025 GC blog introduced you to carrier gases and gas control modes employed in gas chromatography technique. It is never easy subject and becomes even more complex when considering carrier gas type and cost effectiveness of your analysis per sample.
Should you wish to discuss the GC technique and available GC instruments portfolio, please do not hesitate to contact me directly.
RECENT POST
-
Planning a Seamless DWS Installation
Jun 02, 2026
Blog -
Digital Production Quality Monitoring with FreeWeigh....
May 27, 2026
Blog -
Statistical Process Control for Filling and Packaging�...
May 25, 2026
Blog -
Net Content Compliance and Overfill Control in Manufact...
May 05, 2026
Blog -
Building Trust with Legal-for-Trade DWS Systems
May 05, 2026
Blog -
Mason Technology Appointed to HSE National Framework fo...
Apr 27, 2026
News -
Microscopy Technical Training Series
Apr 22, 2026
Videos -
Mason Technology Partners with Azenta Life Sciences to ...
Apr 21, 2026
News
 info@masontec.ie
info@masontec.ie

